The functional attributes of coprocessed excipients in direct compression
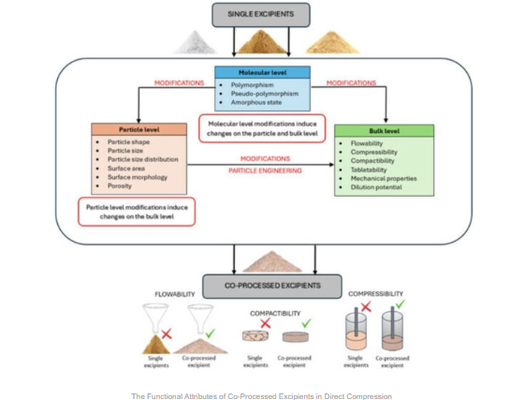
Co‑processed excipients are engineered combinations of two or more individual excipients designed to enhance key powder properties like flowability, compressibility, and compactibility, which are crucial for efficient tablet production—especially direct compression processes. Unlike simple physical blends, co‑processed excipients deliver synergistic performance without significant chemical changes, improving manufacturability and tablet quality. They help address common formulation challenges including moisture and lubricant sensitivity and offer more consistent performance across manufacturing scales. The article also reviews various preparation methods (spray drying, granulation, hot‑melt extrusion, etc.) and how these influence excipient functionality at molecular, particle, and bulk levels. Regulatory considerations, such as the need for risk assessments despite generally safe status, are also discussed.
Comments
No comments posted yet.
















