Highly potent API drug product development and manufacturing using micronization technology
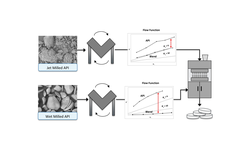
This article covers the critical role of micronization technology in the development and manufacturing of highly potent active pharmaceutical ingredients (HPAPIs). Micronization reduces particle size to improve dissolution, bioavailability, and dose uniformity, which are essential for potent drugs with narrow therapeutic windows. The piece also discusses manufacturing challenges such as containment, operator safety, and process scalability. Advances in micronization equipment and integrated containment systems enable safe, efficient HPAPI production, supporting high-quality drug products with enhanced patient safety and regulatory compliance.

Comments
No comments posted yet.