Quality by design applied to pediatric-friendly formulations
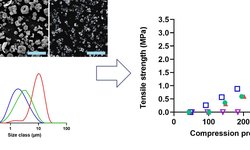
The article explores how Quality by Design (QbD) frameworks can be systematically applied to the development of solid pediatric formulations, with a focus on enhancing safety, efficacy, palatability, and process robustness. Recognizing the stringent limitations on excipient use in pediatric products, the study emphasizes identifying critical quality attributes (CQAs) and critical material/process parameters early in development. This approach often necessitates minimizing or substituting excipients to reduce pediatric exposure while leveraging risk assessment, Design of Experiments (DoE), and taste-masking strategies to deliver practical, child-friendly dosage forms. By mapping these critical variables from the outset, QbD enables more reliable, efficient workflow and ensures pediatric drug products meet quality and regulatory expectations.
Comments
No comments posted yet.