Scientific papers
The amorphous solid dispersion (ASD) is a widely adopted strategy for improving the dissolution of drugs with poor aqueous solubility. However, the selection of an appropriate polymer and drug loading can be a time-consuming process. Surface properties, including surface composition and wetting behavior, play a crucial role in controlling the dissolution of ASD tablets. Therefore, our study aims to utilize surface characterization to gain insights into the factors influencing the dissolution rate of ASD tablets.
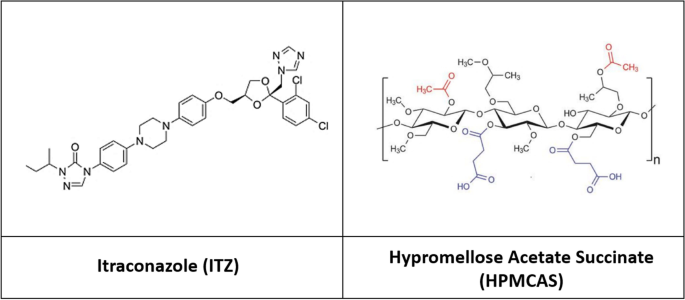
In this study, ASDs were prepared using itraconazole and hypromellose acetate succinate (HPMCAS) via spray drying. Three grades of HPMCAS and various drug loading levels (10%, 30%, and 50%) were employed in the ASD preparation. ASD tablets with two porosities were then fabricated. Contact angles were measured using the Drop Shape Analyzer for each tablet, and surface free energies, disperse, and polar fractions were calculated based on these contact angles. Near-infrared (NIR) and dissolution measurements of ASD tablets were conducted. Principal component analysis (PCA) was performed to further examine the NIR spectra, and relative PCA scores were reported in conjunction with other sample properties.
A partial least square (PLS) model incorporating NIR scores, tablets' wetting properties, and dissolution rates revealed that water and buffer contact angles, surface free energy, and polar fraction are the most influential factors contributing to the dissolution rate of ASD tablets. This study elucidated the intricate relationship between surface properties and the dissolution rate of ASD tablets. Additionally, surface characterization can serve as a valuable tool for screening the formulation and compaction processes of ASD tablets during early development.
Comments
No comments posted yet.
Add a comment