Closing the knowledge gap in high-potency drug development
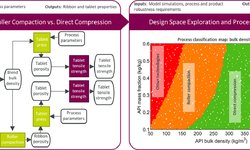
The article “Closing the Knowledge Gap in High‑Potency Drug Development” by Louise Carpenter (Tablets & Capsules, Feb. 2025) explores the growing demand and complexity in HPAPI (high-potency active pharmaceutical ingredient) development, particularly in oncology drug formulation. With nearly 45.5% of small-molecule drug discovery projects involving HPAPIs—and the market expected to grow by almost 9% annually through 2029—pharmaceutical companies face increasing pressure to accelerate high-potency drug manufacturing. Success in this area requires seamless collaboration across formulation development, analytical testing, scale-up, and quality assurance. The article highlights the strategic role of Design of Experiments (DoE) in pharmaceutical development, allowing teams to optimize critical process parameters (CPPs) and critical quality attributes (CQAs) early in the R&D cycle. By leveraging DoE, companies can reduce time-to-market, enhance process robustness, and ensure regulatory compliance when bringing high-potency pharmaceuticals to market.

Comments
No comments posted yet.