Scientific papers
In 1989, Pich and Moest patented mini tablets containing 99.5% pancreatin. Their investigations aimed to tablet the same formulation into 10 mm tablets, but this attempt was unsuccessful. In contrast, Lennartz and Mielck utilized 1.5, 2.3, and 5 mm punches to tablet paracetamol-lactose mixtures, revealing that mini tablets exhibited higher mechanical stability.
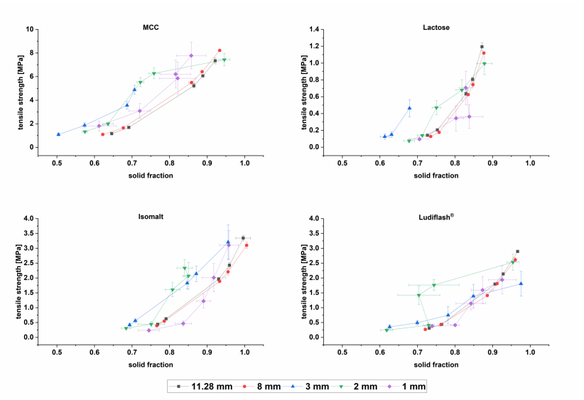
This study delves into the tabletability and compactability properties of four different excipients, examining their dependence on punch size. Microcrystalline Cellulose and lactose were chosen as commonly used excipients in tablet formulation. Additionally, functionalized isomalt and co-processed mannitol were introduced as novel excipients for direct compression. The primary objective of this study is to investigate whether the observations of Pich and Moest, as well as Lennartz and Mielck, were highly influenced by the specific formulation used or if tablet properties are independent of the formulation but reliant on punch diameters. A more systematic and detailed exploration of this topic can be found in the existing literature.
Comments
No comments posted yet.
Add a comment















