Scientific papers
Background
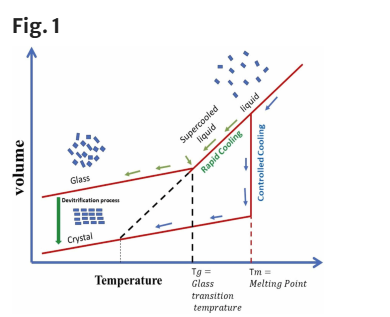
Poor aqueous solubility remains a significant challenge in drug development, limiting dissolution, bioavailability, and ultimately therapeutic performance. Amorphous solid dispersions (ASDs) represent an effective formulation approach to improve solubility and dissolution by promoting supersaturation. However, traditional quality-by-testing (QbT) approaches often lead to inconsistent product quality, increased costs, and longer development timelines. In contrast, the quality-by-design (QbD) framework offers a structured methodology to design robust and scalable ASD formulations while improving efficiency during development.
Area covered
This review explores how QbD principles can be applied to ASD product development. It presents the key components of the QbD approach, reviews relevant quality evaluation methods, and summarizes findings from previous studies. In addition, it discusses tableting considerations for ASDs developed using QbD and examines pharmacokinetic results that can support successful clinical translation.
Expert opinion
Further research should aim to bridge the gap between promising preclinical pharmacokinetic results and clinical outcomes. Key challenges include ensuring scalability, understanding food effects, managing inter-individual variability, and evaluating long-term safety. Expanding the QbD framework beyond manufacturing to include dosage-form development will play an important role in accelerating the clinical adoption of ASD-based formulations.
Comments
No comments posted yet.
Add a comment















