Scientific papers
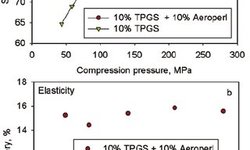
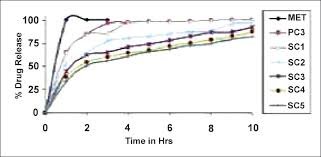
The preparation of lipophilic matrix tablets for the sustained release of water-soluble drugs through direct compression may face challenges due to poor flow and rapid drug release. This study aimed to assess the potential for developing sustained-release diclofenac sodium tablets, utilizing Compritol 888 ATO as a lipid matrix, through a wet granulation technique. The investigation focused on the effects of wet granulation methods (planetary mixer and fluid-bed) and different liquid binders (HPMC Metolose 603, 606, or 615) on weight uniformity, tensile strength, and release rates. Additionally, the impact of compression force and speed during tablet manufacturing under simulated rotary press production conditions was evaluated. Rapid release of diclofenac sodium from directly compressed matrices was observed. However, a wet granulation technique using various HPMC binders resulted in the production of free-flowing granules and matrices that exhibited sustained release of diclofenac sodium over several hours. Further evaluation of the formulation, incorporating the lowest viscosity grade HPMC (Metolose 603), using a laboratory-scale fluid-bed system generated consistently sized granules with good flowability, along with matrices displaying excellent weight uniformity and tensile strengths. Release rates remained consistent across various compression speeds and forces, indicating the formulation's suitability for production on a rotary tablet press.
Comments
No comments posted yet.
Add a comment