Newly published ISPE Good Practice guide
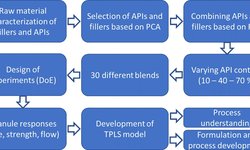
The International Society for Pharmaceutical Engineering (ISPE) has released a new Good Practice Guide focused on developing control strategies for continuous manufacturing (CM) of oral solid dosage forms, including tablets. Continuous manufacturing offers significant advantages over traditional batch processing — such as faster development, greater process robustness, improved quality assurance, and increased operational efficiency. The guide provides a practical framework that combines scientific principles, regulatory expectations, and industry best practices to help manufacturers adopt CM more broadly. It covers key topics such as process model development, integration of spectroscopic techniques, sampling strategies, statistical tools, and regulatory considerations. By documenting the experience of subject-matter experts who have implemented CM in both small and large organizations, the Guide aims to help teams avoid common pitfalls and accelerate the delivery of high-quality, efficiently manufactured oral solid dosage products.

Comments
No comments posted yet.