Aligning pediatric patient needs with drug development success
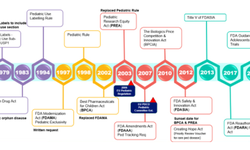
This article discusses the importance of integrating pediatric patient-centric design into the drug development lifecycle to ensure products meet both clinical and commercial requirements. Pediatric formulation presents unique challenges—such as age‑appropriate dosage forms, flexible dosing, swallowability, taste masking, and adherence—that differ significantly from adult development. Regulatory frameworks increasingly require pediatric development planning early in the program, and successful strategies balance technical feasibility with regulatory and market considerations. Collaborations between formulation scientists, clinical teams, regulators, and patient advocates help prioritize pediatric needs, streamline development, and ultimately improve therapeutic outcomes.
Key industry approaches include early identification of pediatric requirements in target product profiles, innovative dosage forms to enhance acceptability and compliance, and cross‑functional alignment to ensure that development decisions support both scientific and patient needs.

Comments
No comments posted yet.