Spray dried dispersions in controlled release formulations
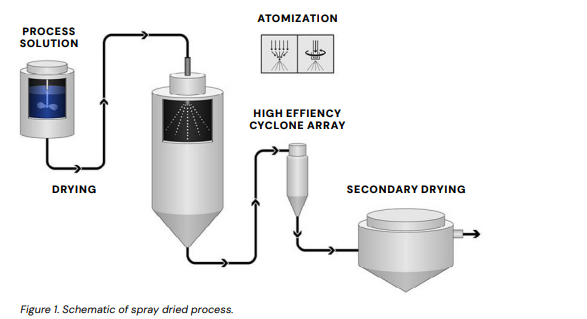
This technical paper examines how spray dried dispersions (SDDs)—a versatile amorphous solid dispersion (ASD) strategy used to enhance solubility and dissolution—can be integrated into controlled‑release (CR) oral dosage forms to address the growing challenge of poorly water‑soluble drugs in new chemical entities. SDDs trap drug molecules in a polymer matrix via rapid solvent evaporation, improving bioavailability and powder characteristics suitable for downstream processing like compression or coating. The document discusses formulation strategies (e.g., SDDs in hydrophilic matrices, coated multiparticulates), the role of polymer selection for both solubility and CR functionality, and practical considerations for development and scale‑up. Although marketed CR products using SDDs are rare, hybrid SDD‑CR systems offer a promising path to combine solubility enhancement with tailored release kinetics for sustained therapeutic outcomes.
Comments
No comments posted yet.