Scientific papers
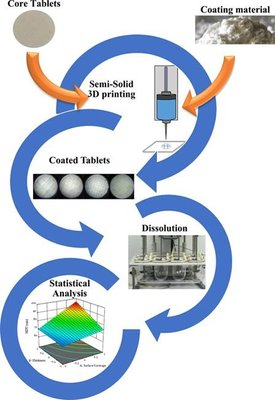
Over the past decade, there has been a growing interest in 3D printing (3DP) technology within the pharmaceutical field, addressing various challenges such as on-demand manufacturing at the point of need, customization of drug release profiles, and the creation of patient-specific solutions. This technology offers a promising avenue for drug product development and manufacturing, supporting specific therapies while enhancing compliance, safety, and effectiveness. This study aimed to partially coat tablets with a glyceride, Precirol ATO 5, utilizing a semi-solids 3D printer to modulate the release of two Active Pharmaceutical Ingredients (APIs): the hydrophilic methyl-levodopa hydrochloride (Melevodopa) and the lipophilic Acyclovir. Through purposeful modifications of various parameters in the 3DP coating process, employing experimental design techniques, the goal was to customize the release profile of the selected APIs without affecting the core composition of the formulation. Key parameters, such as the percentage of the tablet surface coated, the number of coating layers, and the coated sides of the tablet, were systematically adjusted to control the release profile for both APIs. This approach successfully achieved different dissolution profiles by tuning these parameters, revealing a non-Fickian release mechanism for both APIs.
Comments
No comments posted yet.
Add a comment















