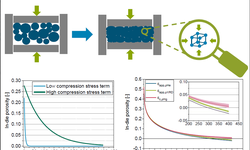
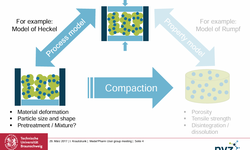
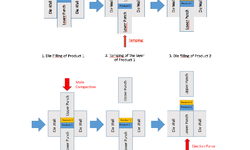
Scientific papers
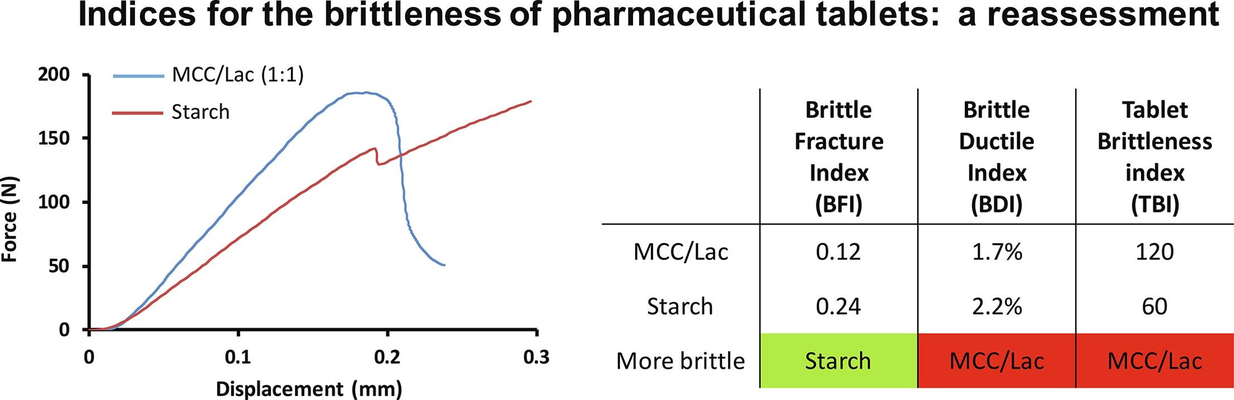
Brittleness stands as a crucial mechanical property, traditionally defined by a material exhibiting elastic behavior until failure under loading. However, it is also sometimes perceived as the material's resistance to breakage. In the context of pharmaceutical tablets, three distinct indices have been introduced to quantify brittleness: the brittle fracture index (BFI), the brittle/ductile index (BDI), and the tablet brittleness index (TBI). This study aimed to reevaluate the interpretation of these indices, which have been known to yield conflicting results. Through theoretical considerations, numerical modeling, and experiments, it was demonstrated that the BFI is the sole index providing an unambiguous measure of tablet brittleness in the classical sense, defined as exhibiting elastic behavior until failure. While the other two indices, such as TBI, may have utility, for instance, in assessing tablet friability, they do not effectively capture tablet brittleness in the conventional sense, i.e., in opposition to ductility.
Comments
No comments posted yet.
Add a comment