Scientific papers
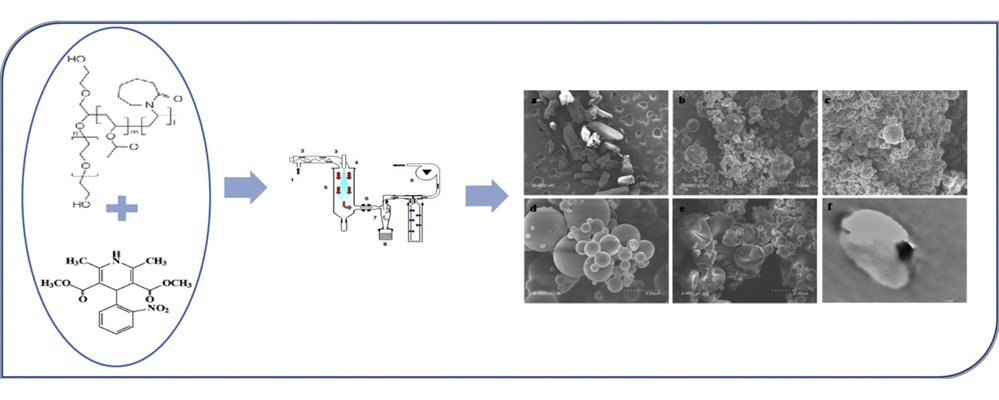
The objective of this study was to explore an alternative processing technology for a novel polymeric solubilizer primarily employed in hot melt extrusion. Poorly soluble nifedipine was co-processed through spray-drying with poly(vinyl caprolactam-co-vinyl acetate-co-ethylene glycol) (PVCVAEG) in varying ratios. The resulting spray-dried powders were then formulated and compacted into tablet forms. Spray drying yielded smoother spherical particles when combined with PVCVAEG, while particles without PVCVAEG exhibited rougher surfaces. Co-processing nifedipine with the polymeric solubilizer led to a reduction in crystallinity. Increasing drug content resulted in the observed plasticization of the polymeric solubilizer. Small-angle diffraction patterns and transmission electron microscopy indicated phase separation throughout the spray-dried particles with high drug content. Compaction with PVCVAEG enhanced the cohesiveness of spray-dried compacts. Heckel modeling demonstrated that deformation of powders containing PVCVAEG was more plastic compared to the brittle nifedipine powders. Dissolution kinetics for all spray-dried samples showed improvement compared to the original nifedipine crystals. However, co-processed nifedipine with PVCVAEG did not exhibit an enhanced dissolution rate when compared to spray-drying nifedipine alone. While PVCVAEG is commonly co-processed with drugs through hot melt extrusion to produce solid dispersions, these results highlight its potential for processing through spray drying to form solid dispersions, with PVCVAEG improving the compactibility of the formulated spray-dried powders.
Comments
No comments posted yet.
Add a comment















