Scientific papers
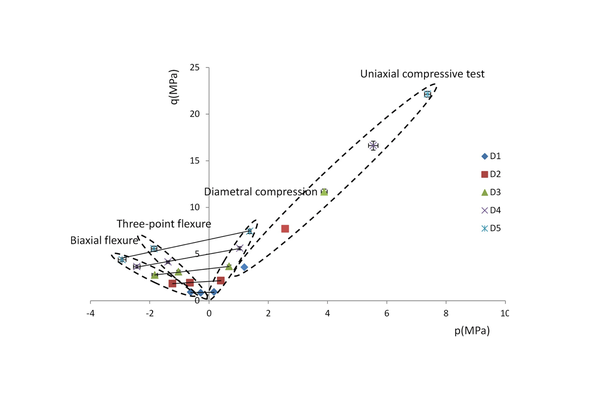
Various tests can be employed to examine the strength of pharmaceutical tablets. Equations are available in the literature to convert the measured failure force into a failure stress, which can be regarded as a material strength characteristic. Each failure test entails a different stress state at failure, resulting in varying failure stresses. Therefore, it would be valuable to identify a failure criterion to unify these diverse results. In this study, four distinct tests were conducted on pharmaceutical compacts with different densities: diametral compression, three-point flexure, biaxial flexure, and uniaxial compressive tests. The Drucker–Prager criterion was evaluated as a potential fracture envelope. The findings indicated that this criterion effectively explains the failures observed in diametral compression, three-point flexure, and biaxial flexure. However, for the uniaxial compressive test, using this criterion led to a significant underestimation of the experimental value of the failure stress. Consequently, the Drucker–Prager criterion should be approached cautiously, as it may not comprehensively explain all failures that can occur in a pharmaceutical compact.
Comments
No comments posted yet.
Add a comment