Scientific papers
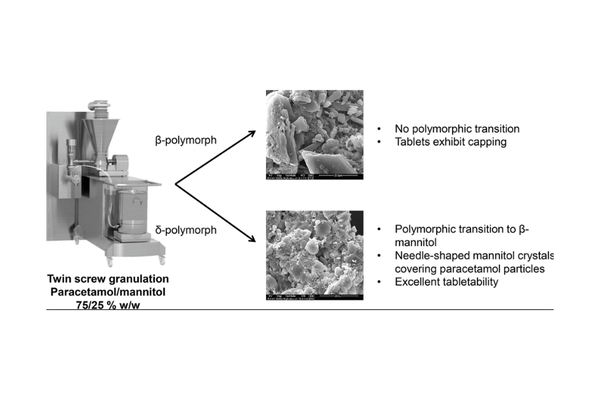
In this present study, we explored whether the moisture-mediated polymorphic transition from δ- to β-mannitol observed during twin screw granulation (TSG) could occur in formulations with a high drug load. Additionally, we investigated if the distinctive granule morphology associated with this polymorphic transition could facilitate the tableting of granules containing 75% paracetamol, a drug known for its poor compactability. The experiments were conducted on an integrated continuous manufacturing line, encompassing a twin screw granulator, fluid bed dryer, mill, and tablet press.
The polymorphic transition from δ- to β-mannitol was indeed observed during twin screw granulation, resulting in granules displaying the characteristic needle-shaped morphology associated with this transition. TSG performed at low liquid-to-solid (L/S) ratios and the use of binders such as polyvinylpyrrolidone or hydroxypropylmethylcellulose were found to inhibit the polymorphic transition. However, factors such as screw speed, drying time, drying temperature, and airflow did not impact the solid state of mannitol in the granules.
Even without the use of a binder, and despite the high paracetamol drug load in the formulation, we observed limited breakage and attrition during the drying and milling processes. Unlike granules manufactured from a formulation containing paracetamol/β-mannitol, which couldn't be tableted due to extensive capping issues, granules prepared from a paracetamol/δ-mannitol formulation exhibited good tabletability. In conclusion, δ-mannitol proves to be a promising excipient for TSG, particularly in high drug-loaded formulations with challenging tabletability.
Comments
No comments posted yet.
Add a comment